SALT LAKE CITY — As scientists inch closer to a potential COVID-19 vaccine, doctors, researchers and public health officials are on their own related lifesaving quest: deciding who will get the vaccine first.
While the federal administration has promised 300 million doses by January 2021, experts are anticipating a much smaller initial supply and have been discussing for months, most recently on Wednesday, how to distribute the first vaccines in a way that’s both protective and fair.
And like most things associated with COVID-19, it’s not easy.
There are nearly 20 million health care providers in the United States, 60 to 80 million essential workers and more than 100 million Americans with underlying medical conditions that increase their risk for a severe or fatal COVID-19 infection.
On top of that, roughly 53 million Americans are 65 or older and at greater risk of suffering from the coronavirus.
So, if the first batch of vaccine doses is 20 million — who gets it?
Complicated though that decision may be, it isn’t the most difficult part of the process.
“There’s always an assumption that if you make (a vaccine) they will come,” said Vicki Freimuth, a former communications director for the Centers for Disease Control and Prevention and a professor emerita in the department of communication studies at the University of Georgia. “We have plenty of evidence that that is not the case, and I think we’re facing a situation where that problem could be worse than normal.”
Public health experts are watching with growing alarm the tidal waves of misinformation and politicization crashing onto the national conversation, causing distrust in a vaccine that isn’t even approved yet.
Consider these facts:
- Only 45% of Americans 18 and over got the flu vaccine in 2019, despite it being a known, safe vaccine.
- 31% of Americans are unsure about a COVID-19 vaccine and 20% said they wouldn’t get one, according to a recent Associated Press-NORC at the University of Chicago survey.
- The measles vaccine was introduced in 1968 and by 2000 the disease was declared eliminated in the United States; however in 2019, the country recorded more than 1,200 cases, primarily among unvaccinated people — the greatest single number of cases since 1992.
While vaccine hesitancy is nothing new, it carries greater weight amid a raging pandemic — more so than brushing aside pediatrician recommendations or forgoing an annual flu shot.
Because no matter how deeply funded, how carefully researched or how equitably allocated a new COVID-19 vaccine is, it has zero impact if people refuse to take it, says Daniel Salmon, director of the Institute for Vaccine Safety within the Johns Hopkins Bloomberg School of Public Health.
“Vaccines don’t save lives,” Salmon says pointedly. “Vaccinations save lives.”
How close are we to a vaccine?
There are more than 170 vaccines in preclinical stages, with eight vaccine candidates in phase III trials around the world — the final step involving tens of thousands of volunteers.
In the United States, three of the six government-supported vaccine candidates are in phase III trials, including Moderna’s and Pfizer/BioNTech’s candidates, both mRNA vaccines, as well as the University of Oxford and AstraZeneca’s viral-vector vaccine.
As labs around the country return phase III findings, researchers will see a bigger picture about vaccine efficacy and any large-scale safety issues — necessary steps to gain emergency use authorization and eventual FDA licensure. (Russia has been criticized for approving a vaccine candidate without a phase III trial.)
Yet despite the title “Operation Warp Speed,” (which experts acknowledge has done little to quell fears of a rushed process) and the unprecedented global teamwork, it’s still possible that all candidates could fail, said Dr. Richard Besser, former acting director of the CDC and now president and CEO of the Robert Wood Johnson Foundation, which is why he uses the word “if” not “when” when talking about a vaccine.
Even if one does come soon, its arrival will not immediately end the pandemic, he says. Instead, it will put the country on a path to recovery — a path which will still include masks and social distancing for a while longer.
Who will get one first?
If a vaccine is identified, those at highest risk will be first in line — but right now that group includes more than half the country.
If you’re under 65 without underlying health conditions and can work from home, this likely doesn’t mean you.
During Wednesday’s meeting of the CDC’s Advisory Committee on Immunization Practices, group members got a visual lineup — blue circles of various shades forming a lopsided Venn diagram.
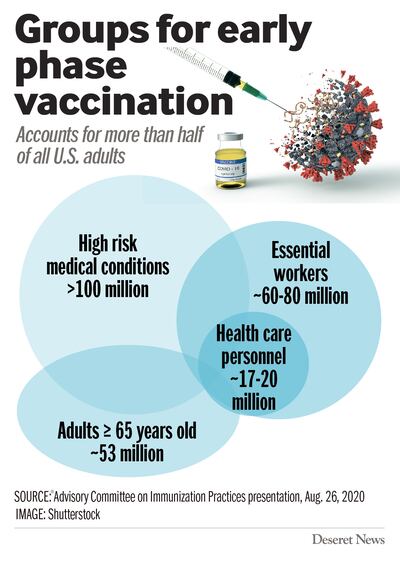
Health care personnel were front and center, with essential workers forming a bigger circle beyond them. Americans with health conditions was a large circle connected to the other side, while Americans 65-plus was a circle on the bottom — all circles overlapping in some way.
While an official advisory committee recommendation isn’t expected until September, the group’s initial consensus was that health care workers should stay top priority, but more sub-categorization is needed.
Picking a second group wasn’t as clear.
“Essential workers and older Americans are almost the same size,” committee member Dr. Peter Szilagyi said on the conference call. “I’m struggling with all of these three circles.”
Szilagyi, a pediatric health services and clinical researcher at UCLA, noted that many essential workers are minorities, who also have experienced significantly higher rates of infection and death from COVID-19, and may also have high-risk medical conditions, making them an essential population to protect.
In nursing homes, where COVID-19 has ravaged both residents and staff, 26% of workers are Black.
Yet, in a mathematical model where Americans over age 65 were vaccinated with a vaccine assumed to be 70% effective, researchers reported a minimal decrease in population-wide infections, but a significant decrease in the number of COVID-19 deaths — more so than vaccinating essential workers or those with underlying health conditions.
Another model showed that vaccinating health care providers in nursing homes decreased COVID-19 cases and deaths significantly more than vaccinating only residents.
Unlike the 2009 H1N1 pandemic, children and pregnant women are not currently considered priority groups and more research is needed to determine whether they should be vaccinated.
What other factors could affect the order?
Allocation isn’t just about ethics and equity — it’s also about freezers.
Two of the vaccine candidates require ultra-low temperatures to remain stable — one at least minus-70 degrees Celsius (minus-94 degrees Fahrenheit). For comparison, a household freezer is around minus-18 degrees Celsius.
“In the first weeks of vaccines being available, implementation concerns are going to be huge,” advisory committee member Dr. Grace Lee, a professor of pediatrics (infectious diseases) at the Lucile Salter Packard Children’s Hospital in California, said Wednesday. “I do think I am supportive of the idea of prioritizing populations where we can actually implement the vaccine.”
Logistically speaking, that would be health care workers, as large hospitals and research facilities will likely have such freezers. Nursing homes must also be prioritized, the committee said, as they’ll likely lack freezer access.
Despite pharmaceutical-company packing solutions with insulated boxes and dry ice, the ultra-low temperature requirements are still a major concern for rural states, said advisory chairman Dr. José Romero, chief medical officer for the Arkansas Department of Health.
He worries that populations like essential agricultural workers may have to wait for a vaccine candidate that’s stable around room temperature.
Given the growing vaccine hesitance, if health care providers — whose vaccine acceptance rate is usually higher than the general public’s — take it first, it could “start a nice process toward introducing confidence in the vaccine,” Szilagyi said.
When will everyone else get one?
For those not at highest risk, it’s more of a waiting game.
But whether the “high demand, low supply” lasts for three weeks or three months is unknown — especially because the initial dose volume remains a mystery.
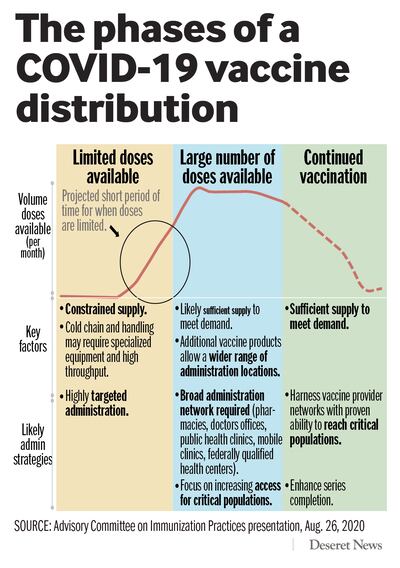
Whenever a vaccine is approved, the administration has announced it will work with McKesson Corp. for distribution. The company notes it currently delivers one-third of the pharmaceuticals used in North America. In prior pandemics, the government has allocated vaccines to states proportional to their population.
After state officials reach those at highest risk, they’ll be able to offer it to the public in traditional places like doctors offices, health clinics and pharmacies.
Experts also hope there’s talk of using nontraditional sites like grocery stores, churches or senior citizen centers that are “convenient, familiar, and feel safe to many vulnerable and underrepresented populations,” according to recommendations from a Johns Hopkins Center for Health Security report.
Four states and one city are working on joint-planning missions with the CDC to navigate what distribution could look like — especially given COVID-19-related restrictions on gathering.
Whenever a federal distribution plan is released, experts insist it must be clear, organized and transparent.
“Otherwise you could have different individuals, organizations and states vying for the vaccine,” said Bruce Y. Lee, a professor of health policy and management at City University of New York. “This was a problem with N95 masks when states ended up having to negotiate their own deals and potentially negotiating against each other, which could drive up the costs.”
How much will it cost?
The United States government has already invested around $10 billion in vaccine development — essentially buying 2.8 billion potential doses of vaccines from six different companies at a price range of $4 to $20 a dose — of which some vaccines need two, according to a recent analysis by AEI.
Yet it’s still fuzzy how much of that cost will be passed to “the people who have to get the vaccine versus the government, which, of course, is taxpayer money,” says Lee.

The administration said a vaccine will be free for those who can’t afford it, and under the CARES Act, any future COVID-19 vaccination was labeled a preventative health measure, meaning it would be covered under health insurance without a copay or deductible.
Insurers are incentivized to pay for the vaccine, said Lee, given that medical costs for a symptomatic person with COVID-19 costs a median of $3,045 — four times more than influenza treatment and 5.5 times more expensive than a symptomatic case of pertussis.
But insurance or not, experts insist money cannot become a hurdle to getting the vaccine.
“Price should not be an issue,” Lee said. “If we have a situation where people can’t get the vaccine, can’t afford it, it will be a huge government and public health failure.”
Is the vaccine process being politicized?
To ensure safe and effective vaccines, the No. 1 rule is to “leave the technical experts alone,” said Alta Charo, a professor of law and bioethics at the University of Wisconsin-Madison.
Those experts include the Advisory Committee on Immunization Practices, a group of 15 medical and public health experts who review licensed vaccines and make recommendations, plus 38 nonvoting members. Without a licensed COVID-19 vaccine yet, the group is focusing on allocation order, sharing recommendations with the CDC.
The National Academies of Sciences, Engineering and Medicine — nongovernmental, nonprofits — also formed a COVID-19 workgroup that will provide recommendations this fall.
However, Charo and Besser are both concerned that political pressure is increasing because of the election year.
- The CDC has already been noticeably absent from the helm of pandemic response, said Besser, with most communication coming instead from the White House.
- On Sunday, Aug. 23, the FDA announced that convalescent plasma had been granted emergency use authorization as a COVID-19 treatment, despite concerns it needed more evaluation. The announcement came one day after a tweet from President Donald Trump in which he called FDA officials part of the “deep state” intent on delaying the research process.
- Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said he would resign if a vaccine was approved by the administration before safety and efficacy was proven, and told Reuters his departure would signal to the country that something was wrong.
The CDC and FDA are world-respected institutions, says Besser, but if they’re not allowed to do their job in an unfettered way, “the public won’t have the same confidence that (their recommendations) represents the best public health science, if it looks like there are political fingerprints overall over it.”
Will the government force me to take it?
The short answer is no. At least not yet.
If an initial COVID-19 vaccine is granted emergency use authorization — instead of full FDA licensure — that version is not allowed to be mandatory, Dr. Amanda Cohn, executive secretary of the ACIP committee, said Wednesday during the meeting.
But here’s the longer answer, and why some licensed vaccines can be mandatory.
In 1902, as a smallpox epidemic raged in Cambridge, Massachusetts, the city’s board of health began to enforce a statute allowing them to require vaccinations of all adults “when necessary for public health or safety.”
Swedish minister Henning Jacobson refused, arguing that he and his son had reacted badly to a prior vaccination. He was fined $5 (roughly $150 today), but refused to pay and appealed to the Supreme Court.
Siding with the state, the court opined that “the liberty secured by the Constitution of the United States to every person within its jurisdiction does not import an absolute right in each person to be, at all times and in all circumstances, wholly freed from restraint. There are manifold restraints to which every person is necessarily subject for the common good.”
That ruling, subsequently called one of the “most important pieces of public health jurisprudence,” has been upheld numerous times, said Charo.
Its impacts are felt every fall, as children show proof of current immunizations (or have a doctor’s note of exemption) to attend public school. States can also require certain vaccinations as a condition of employment.
Yet just like Jacobson (who never got vaccinated, but eventually paid the fine) even mandated kindergarten vaccines does not mean the government is “shoving the needle in your arm,” Charo said.
“It doesn’t force someone to take the vaccine, it just means certain things are no longer available to you if you’re not vaccinated,” she said. “For the sake of everyone’s safety, if you choose not to (be vaccinated) there are certain things you are not permitted to do, it’s just too dangerous to the rest of us.”
How can I trust a vaccine developed so fast?
For experts in public health, the absolute worst-case scenario would be to rush a vaccine to the public that’s neither safe nor effective, said Besser, the former CDC official. Even a vaccine that’s safe, but unhelpful, does little to build or strengthen public trust in the country’s vaccine program.
Which is why, despite the unprecedented speed, experts have repeatedly noted that all safety steps are still in place.

What’s been rushed are the other steps like gathering funding, writing reports and working with manufacturers, said Dr. Richard Rupp, director of clinical trials and clinical research for the Sealy Institute for Vaccine Sciences at the University of Texas Medical Branch, who is conducing clinical trials for both the Moderna and the Pfizer candidates.
Unlike other vaccines that are produced only after FDA approval, promising COVID-19 vaccines are already being mass produced. Companies are gambling that approval will come, allowing them to be ready for immediate delivery.
If they’re wrong, all the doses get thrown away, said Rupp.
These “first-generation vaccines” will have passed the required safety hurdles and antibody measures, said Rupp, but experts won’t yet know if you’ll need a shot every year, like the flu, or every 10 years like tetanus.
It’s possible that “second-generation” vaccines may be more effective, but Rupp said he wouldn’t hold out for a better option. “Personally, I’d get the first vaccine licensed to protect myself.”
Not everyone is quite so eager, but caution shouldn’t be misinterpreted as antagonism or anti-vaxer beliefs.
“It’s completely normal and healthy to have questions about a vaccine right now,” said Kelly L. Moore, associate director for immunization education at the Immunization Action Coalition. “I have questions right now.”
“Our job on the public health side is to get the answers and share these answers with the public so that they’re truly partners with us in this program to protect the world from this COVID-19 pandemic,” she said. “If they know what to expect then they’re much more likely to work with us.”
What about the rest of the world?
Amid all the talk about priority groups, vaccine pricing and public health outreach, experts note this is still a global problem requiring global solutions.
“Only one part of this puzzle is the vaccination policy within the U.S.,” said Charo. “The other part is making sure that there’s adequate supplies for vaccination around the world, because viruses don’t know about borders.”
To ensure every country has access to a vaccine, especially the poorer ones, the World Health Organization, GAVI and the Coalition for Epidemic Preparedness Innovations have set up a vaccine program called COVAX.
Currently, there are 172 countries — 80 higher-income countries, not including the U.S., and 92 low- and middle-income countries — interested in participating in the COVAX initiative, which “works by pooling financial resources to develop vaccines, purchase them at scale, and investing up-front in manufacturing so that vaccines are ready to be distributed as soon as they are licensed,” according to their website.
The goal? Deliver 2 billion doses of safe, effective, approved or WHO pre-qualified vaccine by the end of 2021.
After all, the motto of the initiative is: “No one is safe unless everyone is safe.”




