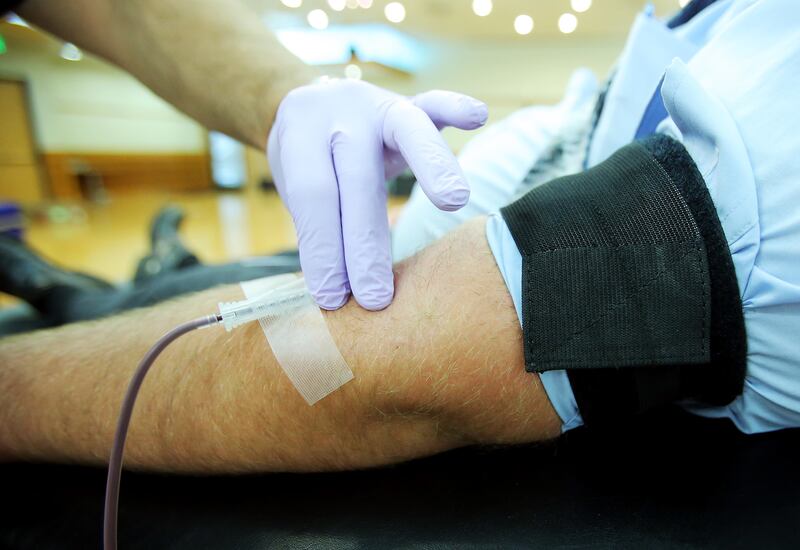
A doctor at the Mount Sinai Hospital in New York is calling for blood plasma from people who have recovered from the COVID-19 virus, hoping to use it to give those who are still fighting the virus a better chance at surviving, the New York Post reports.
The treatment, called “convalescent plasma” would be experimental, and the first of its kind for the coronavirus in the United States, but it was approved by the FDA on Tuesday according to MarketWatch. Convalescent plasma has been used to treat diseases in the past, NBC Washington reports, including SARS and Ebola.
“The idea of convalescent plasma is that people who have had a disease have made antibodies, which are molecules we use to protect ourselves from viruses,” Dr. Bruce Sachais of the New York Blood Center told MarketWatch. “That (plasma) could be collected and given to a person who’s actively sick now … (and) that may help their body begin to fight the infection until their own body makes enough antibodies to clear the infection.”
Eligible donors will be heavily screened, needing a confirmed positive test result, no symptoms for 14 days, a more recent confirmed negative test result and to be found to have high levels of the desired antibodies during a blood test, the New York Times reports.
The experimental treatment would then infuse those antibodies into the blood of hospitalized COVID-19 patients who are moderately affected by disease. Those who are already at advanced stages would not be eligible.