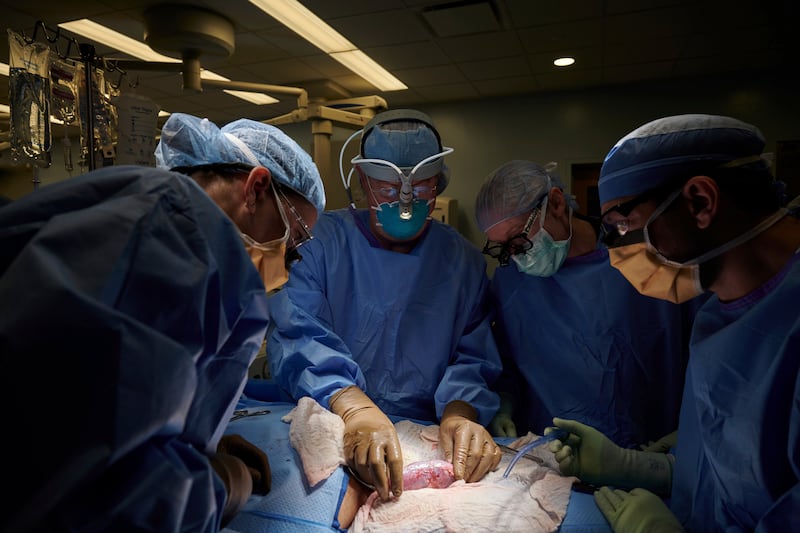
Groundbreaking surgery at a New York City hospital that attached a kidney from a genetically altered pig to a human was made possible by the Nobel Prize-winning gene targeting discoveries of University of Utah researcher Mario Capecchi.
Capecchi pioneered a technology known as “knockout mice” that revolutionized genetic and biomedical research, said Dr. Jeff Campsen, adult and pediatric kidney transplant specialist for University of Utah Health and Intermountain Primary Children’s Hospital.
The procedure performed at NYU Langone Health in September involved a pig whose genes had been altered so that its tissues no longer contained a molecule known to trigger almost immediate rejection.
“What these guys did was they took that technology, that was really created here in Utah, and applied it to this pig. They were able to knock out a gene that’s thought to be one of the major precursors to rejection,” Campsen said.
The recipient was a brain-dead patient with signs of kidney dysfunction whose family consented to the experiment before she was due to be taken off of life support, researchers told Reuters.
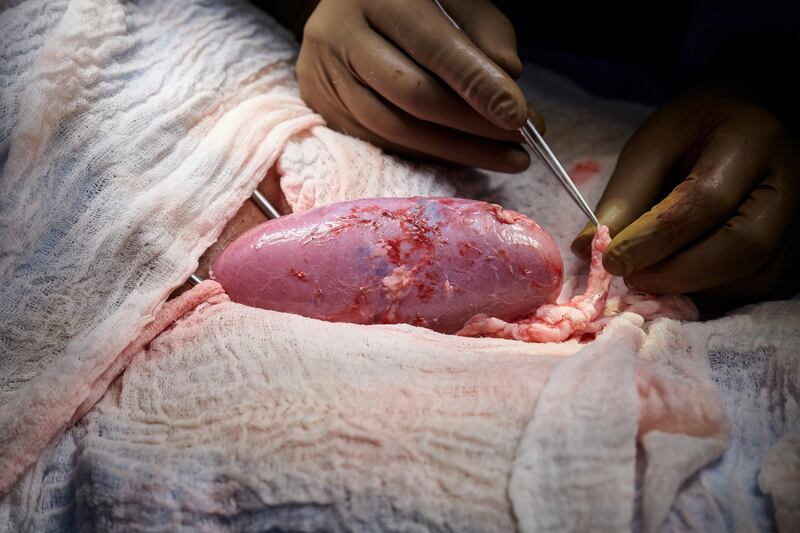
Surgeons attached the porcine kidney to a pair of large blood vessels outside of the woman’s body so they could observe it for two days. The kidney did what it was supposed to do — filter waste and produce urine — and didn’t trigger rejection.
“It had absolutely normal function,” said Dr. Robert Montgomery, who led the surgical team last month at NYU Langone Health, in published reports. “It didn’t have this immediate rejection that we have worried about.”
Campsen described the procedure as “proof of concept,” meaning more research is needed to fully explore the potential. More than 107,000 people in the United States are on the waiting list for an organ transplant, according to the Mayo Clinic. About 20 people die each day awaiting a lifesaving transplant.
“I haven’t talked to those guys but what will be very interesting is what the pathology shows. After they removed the kidney, and then put it underneath a microscope, what markers of rejection did they see?” he said.
A pig kidney is roughly the same size as a human kidney, Campsen said.
Campsen said the experiment was “monumental” in the respect “that they were able to create an experiment that was ethically sound but still doing xenotransplantation into a human being and then getting data from that,” he said. Xenotransplantation means grafting or transplanting organs or tissues between members of different species.
“I do think this was a major advancement because the data that they can glean from this, then can set up a bunch of studies that can then move forward,” he said.
While the work of the NYU Langone Health transplant team “is not conceptually new. It’s sort of the holy grail of transplantation in the sense that can you basically grow your organs, and then just when you’re ready for them, transplant them into the patient that needs to be transplanted?”
Campsen said it is also notable that Montgomery is himself a transplant recipient. He received a transplant of a donor’s hepatitis C-positive heart in 2018. Montgomery tested positive for the disease five days after the surgery and took oral medication every day for eight weeks, and the infection cleared. He was able to return to work full time within three months.