With doctors gearing up for what could be a fifth wave of COVID-19 fueled by the highly transmissible omicron variant, some are warning that several treatments for the virus could be ineffective against the new strain.
Monoclonal antibody treatments have proven to be effective in fighting COVID-19, especially as the delta variant became the country’s dominant strain.
Omicron could change that.
Why omicron treatment options are limited
Dr. Brandon Webb, an infectious disease physician at Intermountain Healthcare, said monoclonal antibody treatments were up to 80% effective at reducing the likelihood of hospitalization or death.
“But with omicron, the variant has a number of mutations in the spike protein that are in the same areas where those monoclonal antibodies target. And the result is that two of our three currently authorized monoclonal antibodies are likely not to work at all,” he said during a news conference. “They’re likely to be completely ineffective against omicron.”
Omicron’s ability to escape immunity applies to monoclonal antibodies, Webb said, leaving doctors with “fewer tools in our toolbox to treat patients.”
One monoclonal antibody treatment — Sotrovimab — is still effective against omicron. But Webb says it’s in a very limited supply and Intermountain is not receiving any allocations of the drug from the federal government.
“We’re in a situation now looking ahead where monoclonal antibodies are likely to be less available,” he told reporters.
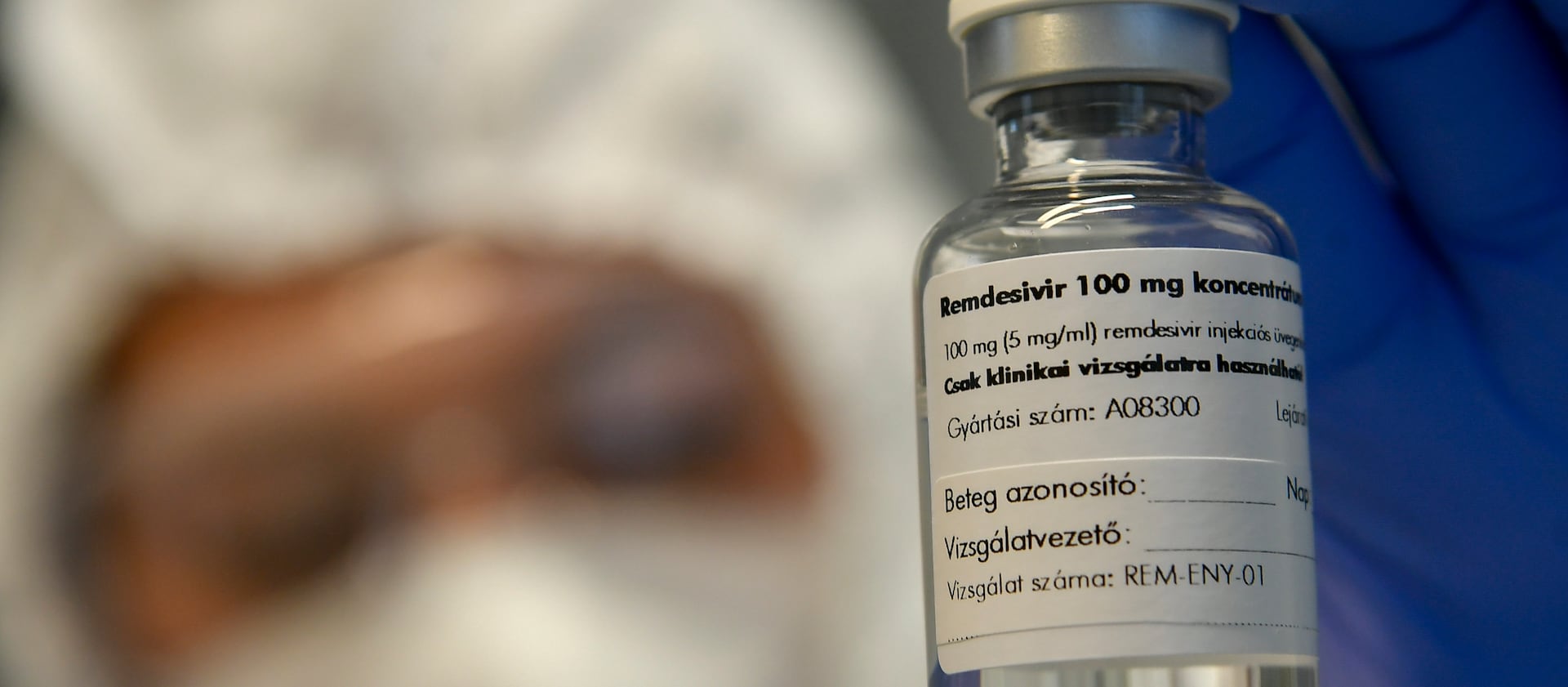
Remdesivir, other treatments, show promise
On Friday, Webb pointed to research set to publish this week that shows the drug remdesivir is an effective treatment against COVID-19 when used early on.
If patients are given a three-day infusion within the first seven days of symptoms, it reduces the likelihood of hospitalization by over 80%.
The drug gained notoriety in 2020 when former President Donald Trump reportedly took it after he contracted COVID-19.
“If you target patients with anti-virals within the first week of symptoms, the effect of the virus itself can be tempered before it causes damage to the organs,” he said.
Webb said as the drug becomes more available to hospitals, it could supplement the waning effectiveness of monoclonal antibodies.
Webb also pointed to two pills he said could be used as treatments for patients with the omicron variant of COVID-19: Paxlovid and molnupiravir.
Produced by Pfizer, Webb says Paxlovid has shown to be more than 80% effective in preventing hospitalization for high risk individuals.
The caveat is that it needs to be used within the first five days of showing symptoms, meaning testing is an important piece to its effectiveness. Webb says it also has 600 drug interactions and certain blood thinners and heart medication will deem some patients ineligible.
The other drug, molnupiravir, has been shown in some instances to be highly effective in treating the virus among unvaccinated patients and is being considered by the FDA for authorization.
Webb says the drug works by introducing a defective building block into the genetic code of the virus. But in some cases, it can cause mutations in the genetic code of the host as well.
“There were some very important reproductive safety concerns that were raised by the FDA,” he said.
Webb also said the drug was tested in countries with vastly different health care systems, and if the drug is approved, it remains to be seen if its success would translate to the U.S.
“At best, that particular drug has modest effectiveness, and in a U.S. health care environment, may have very poor effectiveness,” he said. “So of the two, the Paxlovid drug looks like it will have a more important role in a public health strategy.”