Utah joined other states Tuesday in pausing the administration of the single-dose Johnson & Johnson COVID-19 vaccine while federal health officials investigate reports of rare but treatable blood clots experienced by a small number of vaccine recipients.
Of 6.8 million doses of the Johnson & Johnson vaccine administered in the United States, “a rare and severe type of blood clot” was reported in six women, 18 to 48, whose symptoms occurred six to 13 days after getting the shot, the Centers for Disease Control and Prevention and Food and Drug Administration said in a joint statement.
Although the statement said “right now, these adverse events appear to be extremely rare,” the pause in administering the vaccine was recommended out of what was termed an abundance of caution while the cases are reviewed.
One of the six women who had the blood clots died and a second woman in Nebraska has been hospitalized in critical condition, the New York Times reported.
In Utah, 81,768 people have received the Johnson & Johnson vaccine as of Tuesday, and the Utah Department of Health said none have reported blood clots.
The White House recommended a pause on the vaccine early Tuesday after learning of the cases, said Jeff Zients, White House COVID-19 response coordinator.
Zients said the change will not have a large impact on the nation’s vaccination efforts as less than 5% of recipients so far have received the Johnson & Johnson vaccine.
But Rich Lakin, the state health department’s immunization director, told KSL Newsradio he fears the situation will “scare some people from receiving the vaccine. However, you need to remember this is only 6 people out of 6.8 million doses given.”
Lakin said while those cases need to be investigated, “we still believe that it is a safe and effective vaccine.”
Despite the low number of reports and the fact that it will “slow” the state’s vaccination efforts, Rich Saunders, state health department executive director, called the halt on administering the Johnson & Johnson vaccine “the right thing to do.”
“It’s critical the public be confident in the COVID-19 vaccines, and in order to build and maintain that confidence reports like these must be taken seriously and fully investigated to determine what role, if any, the vaccine played,” Saunders said.
State health officials say they will work closely with the federal agencies to “determine how to move forward based” on their review of the situation and stressed the two other COVID-19 vaccines approved for use in the United States, from Pfizer and Moderna, make up the “significant majority” of doses administered in Utah.
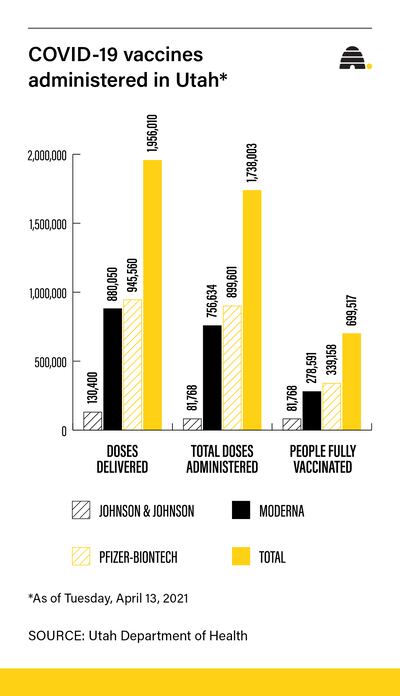
According to Utah’s coronavirus.utah.gov website, Utah has administered more than 1.7 million vaccine doses, including 899,601 of Pfizer and 756,634 of Moderna. Unlike Johnson & Johnson, which requires only a single dose, both Pfizer and Moderna require two doses. Just under 700,000 Utahns are fully vaccinated.
Utah and other states were already expecting fewer doses of the Johnson & Johnson vaccine this week after a recent mix-up at a manufacturing plant ruined up to 15 million doses.
The state health department said Friday Utah was set to receive just 4,900 Johnson & Johnson doses compared to 40,700 last week. No reason was given for the drop, but the state was warned “allocations will be volatile for the next several weeks. The manufacturing process is extremely complex.”
A different type of vaccine
University of Utah Health officials said Tuesday the system “immediately” paused giving all doses of the Johnson & Johnson vaccine. Patients who were scheduled for that vaccine are being offered the Pfizer vaccine instead, said Dr. Richard Orlandi, U. Health chief medical officer of ambulatory health.
The university health system has used the vaccine in just over 7,000 people, Orlandi told reporters during a virtual news conference.
The Johnson & Johnson vaccine is similar to the AstraZeneca vaccine, but the Pfizer vaccine is a different type of vaccine, Orlandi said, reiterating that it remains safe. Blood clots have also been reported from the AstraZeneca vaccine, which is not approved for use in the United States.
“I think many of us have either been vaccinated ourselves with either the J&J or Pfizer vaccine, or our family members have been. And we will continue to move forward with the Pfizer vaccine. We do feel that it is safe,” Orlandi said.
The blood clot cases health leaders are hearing about from the FDA “are cases where the platelets, the things that cause clotting within the blood, can actually clump together. And it appears there’s a concern about whether the immune reaction to the J&J or AstraZeneca vaccine, the concern is that, could that promote clumping together of the platelets, causing blood clots?”
He said similar blood clots are seen in other rare conditions, “and so this is a known, viewed type reaction. It is very rare, and I think that the FDA and CDC are looking into that more to see if there truly is an association between the two (vaccines), and if the risk is increased,” Orlandi said.
‘We don’t want people to overreact’
Intermountain Healthcare, the region’s largest health care provider, is “working to reschedule individuals with appointments for the J&J vaccine for another option. That might mean they have to go to another location, but we are still encouraging individuals to be vaccinated,” spokesman Lance Madigan said.
Madigan said vaccine availability may be reduced because providers have been asked to pause in using the Johnson & Johnson vaccine. However Utahns should still seek to be vaccinated, because the shots remain “the best protection” against the coronavirus, he said.
“Severe side effects are possible but rare, while severe side effects are higher risk for contracting COVID itself. If you have questions, you should look to good information sources such as the CDC or your physician,” Madigan said.
Dr. Tamara Sheffield, medical director of preventive medicine at Intermountain Healthcare, said the blood clots take at least several days to develop after vaccination and should not be confused with more typical symptoms after a shot, such as achiness.
“Just kind of watch yourself for severe symptoms. The types of things that would indicate a blood clot is severe pain in your leg, severe pain in your abdomen, a difficult time catching your breath — the type where it’s the shortness of breath that it feels hard to breath,” she said, “or a severe, severe headache.”
The CDC is advising that anyone who develops those symptoms after getting the Johnson & Johnson vaccine should contact their health care provider and get evaluated, Sheffield said, adding that it is still not known if the blood clots are caused by the vaccine.
“They’re still investigating because there are so few numbers of cases. But we want to have this abundance of caution and I’m so glad they’re stopping things to reevaluate and to look for the evidence,” she said, anticipating there could be an answer about the vaccine by the end of the week.
Sheffield said the pause has made her “more confident in vaccines because I know the right safety measures are taken around them,” but acknowledged others may not agree.
“Anytime there are safety concerns that are brought up on a vaccine, it will cause people to have a second thought about it,” she said. “People need to be weighing their risks and benefits. But we don’t want people to overreact on this. We want them to be considerate and thoughtful.”
Two more deaths from COVID-19 reported in Utah
The news comes as the state health department is reporting 358 new COVID-19 cases and two additional deaths from the virus. Utah has now had 390,647 coronavirus cases since the pandemic began more than a year ago.
The state inoculated just over 30,000 people since Monday, administering a total of 1,738,003 vaccine doses.
The rolling seven-day average for positive tests is 406 per day, with an additional 5,683 Utahns tested since Monday and 15,222 tests given. The rolling seven-day average for percent positivity of COVID-19 tests is 3.8% when all results are included, and 7.7% when multiple tests by an individual in the past 90 days are excluded.
Currently, there are 124 people hospitalized with the coronavirus in Utah, bringing the state’s total hospitalizations to 15,804.
Utah’s death toll has now reached 2,161 lives lost with the deaths reported Tuesday — a Salt Lake County woman between 45 and 64 who was hospitalized at the time of her death, and a Carbon County woman, between 65 and 84, who was not hospitalized.



