SALT LAKE CITY — The pandemic has created global shortages of critical supplies to protect health care providers and enable COVID-19 testing.
That shortage is influencing decisions about how the viral pandemic health crisis is handled — including who gets tested — driven in part by stocks of masks and gloves and the chemicals to run the tests. And health experts, policymakers and others are pondering ever-more-drastic steps.
Their goal is to broaden tight supply chains to get enough material to protect health care workers and lab techs and to run the tests that verify infection, experts told the Deseret News. Solutions now involve governments, manufacturers, health care providers and even individuals.
The shortages have made a severe crisis even worse, with officials begging for help. For example, people who purchased masks, gloves and other protective equipment for personal use early on are asked — along with primary care doctors, veterinarians, dentists, construction workers and others who might have their own supplies of certain types of masks — to donate them to providers for the public good, said Kevin McCulley, whose titles include Preparedness and Response director for the Utah Department of Health.
Meanwhile, officials are tightly controlling who is tested, limiting it to those with active symptoms and a health care provider referral. Two-swab testing and confirmatory tests have been nearly eliminated to halve the use of supplies.
Now some experts even ponder if homemade face masks could allow health care providers to reuse N95 respirators.
Only drastic measures will beat the virus and the highly contagious respiratory disease that has infected more than 441,187 people worldwide and killed close to 20,000 globally, including infecting more than 55,000 Americans and killing 802 as of Wednesday morning. Experts are challenged by the sheer volume of cases: All but a few countries have lab-confirmed infections and many compete for the same protective gear, chemistry and other materials, creating the shortages.
“COVID-19 is a global issue and therefore global demand for supplies is unprecedented. We are working with suppliers who are doing all they can to produce necessary supplies of equipment and other items. Most supplies are on allocation with ‘hot spots’ getting priority,” said Jess Gomez, an Intermountain Healthcare spokesman.
Laboratories have similar problems.
“The challenge for the global supply chain is that a lot of different labs are running the test and need the same materials. It requires balance so that everyone gets what they need,” said Dr. Brian R. Jackson, a medical director at ARUP Laboratories.
Shoring up supplies
To preserve personal protective equipment for medical professionals dealing with COVID-19, Utah health officials this week announced restrictions on medical, dental and veterinary procedures that are not urgent. Gov. Gary Herbert said the restrictions would run until April 25.
In a written statement, Herbert said he appreciates “the patience of Utahns who were planning procedures that will now be delayed. Although the term ‘elective’ indicates something that is non-essential, I realize this will still be an inconvenience, and for that I am sorry. As we look at the experiences of other states and regions of the world, it’s clear that those who are proactive in securing a supply of (personal protective equipment) are far better equipped when they see a surge of COVID-19 patients being admitted to hospitals.”
The federal Centers for Medicare and Medicaid Service said elective procedures include colonoscopies, cataracts, endoscopies and others that could wait without putting patient health in danger.

McCulley said both industries and individuals are willing to tackle shortages; Utah officials receive hundreds of offers daily to purchase personal protective equipment for the anti-COVID effort and to team with private-sector partners to buy manufacturing products and materials necessary for testing for and treating COVID-19 that are in short supply.
McCulley said officials have to be specific about needs to head off offers like crocheted blankets, which don’t help.
Coronavirus.utah.gov’s donation page asks primary care physicians, dentists, veterinarians and some industries to donate N95 masks, medical-grade shields or goggles, surgical gowns, quality medical gloves and other personal protective equipment to frontline health workers. Protecting health care workers’ faces and hands is the highest priority, though gowns and other gear are also needed, he said.
Utah’s Silicon Slopes technology community is also soliciting donations and volunteers to counter the crisis through its slopesserves.com website, McCulley said.
“Our understanding is that the response from community partners has been impressive,” said Gomez, who noted Intermountain, the Utah Department of Health and other community organizations are working together to obtain supplies.

Degrees of need — and crisis
The CDC released conventional, contingency and crisis care guidance for stretching supplies of personal protective equipment, including masks. When supplies are adequate, there are strict rules about quality, expiration dates and other factors. The rules thin in this kind of pandemic, but drastic crisis management kicks in only when other management efforts don’t yield enough supplies.
Health experts consider reusing some protective equipment that’s usually single use, for instance, McCulley said. Or using expired respirator masks or those that meet other countries’ standards but are not certified to U.S. standards, he added.
Even using homemade masks could be considered if the shortage is dire enough — ”with absolute recommendation for caution when considering that type of an option,” said McCulley. He noted surgical and homemade masks might help extend N95 respirators, perhaps by placing them over the top of the coveted N95 respirators to minimize contamination, then swapping out the overlaid mask so the N95s last longer.
“We recognize that each of these strategies is for consideration when we are absolutely unable to generate any supplies for the preferred, appropriate and recommended use to meet the infection-control standard. Crisis care means that you have to get creative in this. Certainly I would hate to say that I’m recommending that,” McCulley said of home-sewn masks.
Public health experts also worry about the potential that a layered mask could make it hard for a health care provider to breathe; some already find it a bit difficult with just an N95. On the other hand, “layering might extend or allow for alternative use of respiratory protection equipment.” The trick is to minimize risk to the health care worker from using products in ways that they were not intended and designed to work, McCulley added.
Testing needs
Safety practices for handling patient specimens for respiratory infections like the SARS-CoV-2 virus that causes COVID-19 are well established, according to ARUP’s Jackson. “Wearing lab coats, gloves and face shields, using a biosafety cabinet and working with appropriate spacing are crucial safety measures for this type of testing,” he said.
Jackson said ARUP follows “rigorous standards and practices for cleaning, personal hygiene, environmental controls and personal protective safety measures” like restricting entry to the building to those processing the day’s tests.
“The biosafety issues in the lab are no different than those we deal with day to day. COVID requires the same precautions that we typically use for testing. We process specimens with all kinds of infectious agents on a daily basis,” he said.
But one of the factors limiting testing volume is international in scope, due to lack of supplies, including swabs and media to transport them, Jackson said. “We are involved with multiple calls daily with workgroups, FEMA, the American Clinical Laboratory Association and others trying to address, understand and navigate this issue. “
Labs that process COVID-19 tests can’t predict the volume of supplies the pandemic’s testing will require, but most told the Deseret News they have enough to meet the current need.
That wouldn’t be true with a huge increase in cases or if testing expanded to the general population. “It is important that access to testing continues to be prioritized based on clinical guidelines to protect capacity and testing speed for all those in most urgent need,” Jackson said.
Testing also requires equipment like pipettes and chemicals called reagents. And those processing the tests need personal protective equipment.
“The safety of lab personnel is paramount,” McCulley said. “You can’t just stick somebody else in that seat. You have to absolutely protect the scientists in the laboratory as they continue to process this rising number of tests per day.”
Even nasal swabs are closely guarded to ensure an adequate supply.
FastBreak reported that because of test and testing supply shortages, some health care systems, including at Johns Hopkins University, University of Washington and Mayo Clinic, had come up with their own tests for COVID-19, with more expected to follow suit.
A federal response
Congress’ stimulus package reflects national concern with the problem. According to the Chicago Tribune, the agreement includes $100 billion for hospitals and health systems, and billions more to give them more personal and protective equipment for health care workers, testing supplies, among other things. It doesn’t say where the supplies will come from — and that’s the big question with the supply chain shortage.
The Deseret News reported that according to The Hill, “In a letter, attorneys general from 16 states called on the president to invoke the Defense Production Act to direct industries to create more necessary medical supplies to combat the coronavirus pandemic.”
Some companies are already stepping up. In Utah, for example, SugarHouse Industries has begun repurposing vinyl it uses in boat covers to instead make face shields and masks needed in health care.
Other companies could be compelled to help.
President Donald Trump signed the Defense Production Act of 1950, which would let the administration force U.S. manufacturers to produce medical supplies and equipment and divert materials and facilities as needed to deal with shortages. That act could also be used to increase production of chemicals and other supplies used for lab testing. But while national hospital, physician and nursing groups wrote a letter asking Trump to invoke the act, he tweeted he would only implement measures in a “worst-case scenario.”
State and federal health officials, in partnership with others, are looking at less drastic measures to boost critical supplies.
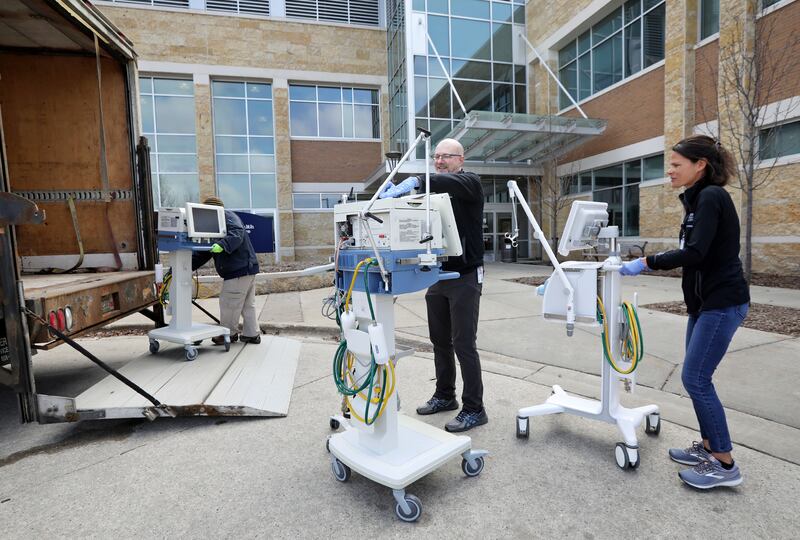
The Food and Drug Administration has agreed to allow modification of ventilators and respiratory equipment and permit use past normal expiration in some cases to increase their availability during the pandemic.
The FDA said measures to extend the supply of gloves can include using nonsterile disposable patient examination gloves designed for routine patient care. Health care providers can train with expired gloves, among other practices. In certain circumstances, gloves could be used on more than one patient with the same infection, if supplies are critically low, the agency noted. Possibilities change periodically to meet the scope of the crisis.
Each state gets a proportional share of the Strategic National Stockpile maintained by the federal government. Utah has already received a modest share of the supplies. Most of the stockpile will go to the hardest-hit states.
Officials must also maintain some supplies for down the road, in case things get much worse. “We have some concerns about exhausting our supply prior to Utah seeing the type of medical surge that states like New York and Washington have seen,” McCulley said. “There’s a fine line between getting stuff into use right now and getting stuff and initiating a conservation strategy so whatever you have, you will still have enough in place to deal with an inpatient surge. We can’t say that’s going to come to Utah, but we have to think about conservation now to ensure enough remains when and if an ICU surge comes.”
He said health care systems don’t usually have much excess capacity — of supplies or space. They operate at a lean, efficient level to keep costs down.
As for the DPA, McCulley said he’d love to see more manufacturing capability to stabilize the shortages, but wonders how long it would take. If factories and manufacturers needed three months to be ready, “that might be a little late for us,” he said.
“I am all in favor of any and all federal efforts to enhance production, manufacturing, sourcing and distribution — anything the federal government can do to support us in the states. But we are not going to wait for a federal option. We have multiple teams working on sourcing, ordering, donation and vetting the opportunities being presented to us right now. We are doing as much as we can and continue to encourage our private sector partners to work within the normal distribution supply chain. Everybody has to keep pushing,” McCulley said.
In the pipeline
Current testing detects the virus directly; antibody tests detect a patient’s immune response to a disease and are useful to evaluate patients who may have had the infection in the past and recovered. Antibody tests could be useful to assess risk of future infection, as well as for public health agencies as they study the epidemic.
They are not yet widely available, but the CDC and others are developing them.
Federal officials have also said they might be useful to determine whether some health care workers who had the disease, whether they knew it or not, carry some immunity to infection and thus could be valuable in treating those with active COVID-19.




