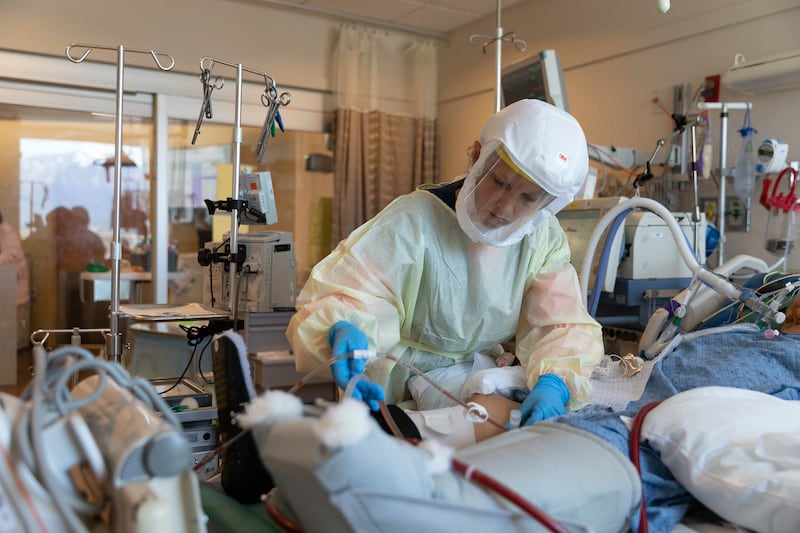
Stumped in finding a way to treat a “devastating” syndrome brought on by COVID-19, doctors and hospitals worldwide are joining a new effort to combat the deadliest damage caused by the novel coronavirus.
Utah hospitals are participating in the National Institutes of Health-sponsored study aimed at finding a much-needed treatment for the “sickest of the sick” COVID-19 patients, an Intermountain Healthcare researcher and critical care physician said Wednesday.
Dr. Samuel Brown said four coronavirus patients with acute respiratory distress syndrome, a life-threatening condition that affects the lungs, have been enrolled in the study at the Intermountain Medical Center in Murray and the University of Utah Hospital in Salt Lake City.
The “devastating” syndrome is the primary cause of death in patients with COVID-19, Brown said. The ACTIV-3 Critical Care trial testing the effectiveness of two drugs, Zyesami and the antiviral remdesivir (Veklury), in treating patients hospitalized with life-threatening cases of COVID-19 was announced last week by the federal government.
“So far, we’ve been struggling to find therapies that will work for the sickest of the sick, the people who are on life support treatments because their lungs are failing,” Brown said. He said the syndrome can kill 20% to 40% of patients and it can take the majority of survivors months or even years to recover, while some never will.

The doctor compared the damage to the lungs when the immune system overreacts to the presence of the virus there to the collateral damage left behind by dropping a massive bomb on a small target rather than conducting a surgical strike.
The delicate structure of the lung ends up clogging the foam-like walls with mucus, dead cells, blood clots, water, pus and inflammation, Brown said.
“You can’t breathe through that. It’s like drowning. Many patients describe the experience of this terrible infection of the lung as suffocating and like drowning.”
Zyesami is believed to protect lung cells attacked by COVID-19 and has shown it can decrease the amount of the virus and adjust the inflammation and immune response, while producing more of a lung-strengthening chemical in the body, he said, while the antiviral remdesivir has already helped in less severe coronavirus cases.
Brown is the principal investigator in the study, also underway at sites throughout the world, including networks based at the University of Minnesota, Massachusetts General Hospital in Boston, Mount Sinai Hospital in New York City and in Washington, D.C., as well as in Denmark, Spain and Brazil.
“We’re very excited,” Brown said. “We’re moving rapidly. Part of what’s so hard about COVID is you want this ... virus to be gone. We want to be done with it. Nothing would make us happier than to never have to do another COVID study for the rest of our lives. The reality though, is that COVID persists.”
As long as that’s happening, he said, “we feel a moral obligation to bring trial access to patients to allow them access to these promising drugs in a scientifically careful and safe and appropriate environment such as you see with a clinical trial, and as quickly as we can, to get answers.”
How long that takes depends on how many patients are in the trial, something that’s been an issue in other studies as case counts drop. Right now, there are about 50 study sites in the United States and another 30 to 40 in other countries. In Utah, patients or their family members can talk with their doctors to see if they qualify to participate.
Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases and chief medical adviser to President Joe Biden, stressed the importance of the study in a recent statement.
“Finding more effective therapeutics for critically ill COVID-19 patients remains an essential need in the ongoing response to the pandemic,” Fauci said. “We need more treatment options to increase the chances of recovery for people who are extremely sick so they can leave the hospital more quickly, continue their recovery at home, and return to life as usual.